Brachytherapy is a method of radiological therapy by which a radiation source penetrates directly in the target tissue of the affected site, the tumor nidus, without interfering with other systems or structures of adjacent soft tissues. To remove a malignant tumor of prostate gland an interstitial brachytherapy is used, which enables to treat not only the prostate gland, but also soft-tissue-organs.
Brachytherapy in Germany is done on a high technological level due to the professionalism of the physicians who use only modern equipment.
With the help of radioactive needles or applicators the source is placed directly into the affected area, minimizing the radiation exposure to adjacent tissues.
There are two methods of prostate brachytherapy, each of them has its special features. The first method involves a temporary radiation source which is removed after the therapy leaving no radioactive material in the prostate gland. The second one implies injection of radioactive seed implants into the prostate gland that remain there permanently. One of the main reasons for brachytherapy in Germany is a low complication rate.
Types of Brachytherapy
Today, brachytherapy is considered to be the most optimal treatment option for patients with cancerous tumors in different organs: cervix and uterus, vagina, prostate gland, esophagus, intestine, eyes, etc. Depending on the medical equipment, tumor location, and anatomical peculiarities of organs and tissues, the following types of brachytherapy exist:
according to localization:
- intracavity (gynecology, proctology);
- interstitial (prostate);
- intraluminal (esophagus, bronchi);
- superficial (application);
- intravascular.
according to the application:
- manual (manual after-loading) – installation and removal of radiation element is done manually.
- automated (remote after-loading) – automated distance sequential load of the element. The source is placed in a special container, and during the procedure, is taken to the planned point of the applicator via entrance channels. After that, it’s taken back to the storage space of the apparatus.
Intracavitary brachytherapy is used when the tumor is located in cavernous organs. Special applicators are injected in patent’s cavity, and then a radiation element is delivered according to a special treatment program. This form of brachytherapy is widespread for treatment of tumors in uterus, vagina, anal channel and intestine. The technology is also used to treat organs with a gap – esophagus, bronchi, bile ducts, etc. Today, intracavitary brachytherapy is done with the help of advanced medical technologies, which allows calculating an optimal dose to treat this or that cancerous tumor.
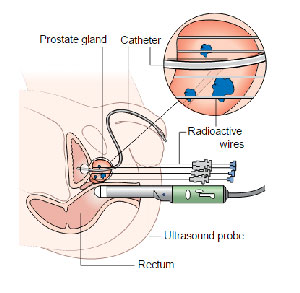
Interstitial brachytherapy is used to treat organs with tissue structure. In this case, the radiation source is injected into the affected tissue with radiation needles or needle-form applicators. Today, this treatment method is mainly used to treat prostate cancer. However, the two existing methods of implantation have certain peculiarities: use of “temporary” sources (most often, iridium source is used), and constant sources (iodine and palladium stay inside patient’s body after treatment). One of the main advantages of interstitial radiation therapy is a low risk of complications, which cannot be applied to distance radiation therapy and surgical treatment.
Intravascular brachytherapy is an active method for treating restenosis of coronary vessels. For that purpose, the source of β- or γ- radiation is used: for some time, it’s integrated into vessel gap. Intravascular brachytherapy is being used all around the world. However, specialists claim that this type of treatment is hard to perform technically, because special medical apparatus should be used, and the medical staff should be protected against radiation properly. Today, scientific research is being performed to make this method more available.
Indications and Contraindications to the Brachytherapy Procedure
The direct, targeted radiation therapy is carried out only in the early stages of the disease ‒ T1 and T2. The reason is that the area of the radiation spread around the affected organ is only 1.5-2 mm.
The following factors are indications to prostate brachytherapy:
- the malignant tumour did not spread through the walls of the prostate;
- the PSA (prostate specific antigen) value is 10–20 ng/ ml;
- the Gleason score is lower than 7;
- the organ along with the tumour is enlarged to 60 cm3.
Failure to comply with these rules will lead to the opposite effect ‒ the lesion focus will start spreading quicker.
The contraindications to the procedure are:
- urinary incontinence, too frequent urination;
- the patient has had a history of transurethral resection of the gland ‒ an absolute contraindication, since after treatment with such irradiation the patient will suffer from urinary incontinence;
- tumours with poor prognosis;
- the use of anesthesia is impossible;
- the prostate is enlarged (more than 60 cm3);
- inflammation in the rectum.
The last items are relative ‒ after treatment for these diseases or having reduced the size of the gland up to 50% by using hormonal therapy, the method becomes acceptable.
Brachytherapy for Prostate Carcinoma
Treatment of prostate carcinoma is one of the relevant problems of modern urology. Today, this is the most widespread cancerous disease among men: it’s the top reason of lethal cases caused by cancer. Thanks to screening program with PSA specification, the number of revealed prostate carcinoma cases is growing from year to year. In 2003, 220.900 of cases were revealed, and 28.900 died from cancer. The type of treatment offered to a patient depends on his age and concomitant diseases. At early stages of cancer, relatively young patients should choose radical prostatectomy (surgical removal of the prostate gland). External-beam radiotherapy and brachytherapy are alternatives to radical prostatectomy. If the tumor is revealed at a late stage, doctors select beam therapy, hormonal therapy, or their combination.
The history of brachytherapy for prostate carcinoma
The history of development and implementation of brachytherapy for prostate carcinoma is pretty interesting. In 1970-s, only a few centers used brachytherapy to treat this disease. Implants were injected into the prostate gland under oncologic surgeon’s supervision during an open cavity operation. This type of treatment caused serious complications and wasn’t efficient, which was conditioned by the technical imperfection of equipment used in those times. That led to imprecise and inefficient implantation of radiation sources. Besides, at those times, there were no objective criteria concerning the dose of radiation applied to the prostate gland. A new wave of interest was in 1980’s when brachytherapy started being used as an addition to exterior beam therapy, and the technique of radical prostatectomy was being developed and improved. At the edge of 1980-1990’s, with the help of transrectal ultrasound (more advanced method of prostate gland visualization) and a plotter that directs implantation needles, the method of percutaneous transperineal brachytherapy was introduced to treat localized forms of prostate cance when there are no metastases, and the tumor hasn’t spread outside the organ yet. Besides, doctors managed to reach higher radiation dosage for the prostate cancer than during exterior radiation therapy and avoid damage of adjacent organs and structures.
Treatment of prostate carcinoma with brachytherapy in our times
Today, brachytherapy is used to treat prostate carcinoma at early stages. Different centers use different methods of radiation source transplantation, but all specialists follow the recommendations of the well-established German society of brachytherapists. First and foremost, these recommendations concern permanent low-dose brachytherapy. These recommendations mention the criteria for patient selection, the definition of low, middle and high-risk groups, optimal dosage of beam radiation, characteristics of isotope types, limits of radiation loading for critical organs, etc.
Brachytherapy for a disease of any location is a multi-disciplinary type of treatment. The German society of brachytherapists recommend organizing surgical teams to perform brachytherapy procedures that should include: a doctor of particular speciality, as a rule, surgeon (urologist, angiosurgeon, cardio surgeon, gynecologist, etc), radiation oncologist, a specialist of medical physics, anesthesiologist, instrument nurse, and a nurse- anaesthetist. All these specialists collaborate closely during all stages of brachytherapy, including preparation, performance, and evaluation of treatment results. That’s why the team should learn collectively and touch upon all basic regulations in medicine.
According to the recommendations of the German society of brachytherapists, radiation-monitoring planning should be performed before or during implantation, and the check date is 4-6 weeks after the procedure. Today, computer tomography is used as the main visualization method, because it’s more widespread, available and efficient way of getting images of prostate and implanted sources.
Other methods of visualization are also applied: ultrasound, MRI, and X-ray inspection are used to define the form, sizes, and location of the prostate gland to control the location of implants inside. The following stages are widely used:
- Pre-implantation planning (Preimplant) several days (not more than 4 weeks) prior to the procedure for dose planning;
- treatment planning during the procedure;
- interactive planning during the procedure using software to correct radiation dose;
- postimplant research 4-6 weeks after the procedure to evaluate the dosage.
Transrectal sonography and computer tomography are used as visualization methods for brachytherapy. Depending on the method and way to obtain images, a certain “target” should be defined during transplantation. This definition includes the following structures: prostate gland with its contours, its top (apex), base, periprostatic fiber, and seminal glands. Additionally, doctors need to visualize the pelvic muscles on Apex level and adjacent tissues on the base level due to possible migration of micro-sources in these zones.
Today, two methods of permanent brachytherapy with constant implants are used: under control of transrectal sonography or computer tomography. When implantation under CT control is performed, scanning for planning is done several days before. For better visualization of prostate gland and neck of the urinary bladder, the X-ray contrast agent is injected via a urethral catheter. Injection of trocar needles for implantation and radioactive sources is controlled by constant CT scanning during the implantation procedure: 4-6 weeks after brachytherapy, CT scanning is done for post-implant dosage calculation.
Even old people with concomitant diseases tolerate brachytherapy well. The next day, a patient can leave the clinic and get back to normal life. 79% of men live at least 10 years without complaints. It’s very positive statistics.
Sources for brachytherapy
Radioactive micro-sources used for brachytherapy and target implantation are closed radiation sources of low activity that have standardized radiation characteristics.
Modern closed sources for interstitial beam therapy are high-tech products. These are titan capsules with dimensions of 4,5 mm х 0,8 mm and wall width of 0,05 mm. Considering the half-life and way of radiation diffusion in tissues, the following isotopes are the most convenient for a clinical appliance: 125I, 103Pd, 131Cs. Radioisotope absorbed on a silver or graphite matrix is located inside the capsule. The ends of the capsules are sealed with a laser. Micro-capsules can be made in the form of “free grains”, or be fixed on a self-dissolving polymer thread. The advantages of thread-fixed sources: no risk of grain migration, the correct geometrical location of grains – precise dosage, quick charging of needles – shorder procedure duration and less effect from radiation to the operator, safety of storage and transportation.
Today, new radioisotopes are being searched for brachytherapy, and new types of micro-sources are being developed. Today, developmental prototypes made of cheaper and modern materials are being elaborated.
How is the Procedure Performed?
Low-dose rate brachytherapy is usually carried out in the outpatient setting; sometimes it takes place in stationary conditions.
Before the procedure, a therapeutic scheme is made, which is the most important stage of the treatment. Its goal is to calculate the radiation power necessary to completely destroy tumour cells.
The following patient examinations are also performed:
- Digital rectal examination through the anus.
- Determining the amount of PSA in the blood, a high value indicates prostate cancer.
- Transrectal ultrasound examination (TRUS) to determine the size and location of the tumour, as well as to develop an optimal plan for the location of radioactive sources. Detailed ultrasound examination is performed within 30-40 minutes. Planning can be carried out both immediately before the operation and several days before it.
- If necessary, instead of TRUS, or in addition to it, the doctor may prescribe a computed tomography scan of the pelvic organs.
- Histological examination of a prostate biopsy from the tumour tissue.
- Assessment of urination (residual urine volume).
The procedure for implanting radiation sources is carried out under ultrasound or fluoroscopy control and includes several steps:
- Performing epidural or spinal anesthesia. In some cases, general anesthesia is performed.
- Positioning the patient on the operating table (supine, with the legs spread at 90 degrees, fixed in the holders).
- Getting ultrasound images with a step of 5 mm in the coordinate grid from the base of the prostate to its top; planning the insertion of radioactive sources. The ultrasound probe is inserted rectally. If the gland is located below the coordinate grid, it is raised a little with a balloon filled with water. The balloon is mounted on a stepping device. For a more clear visualization of the urethra, foamed gel is used, or a gas-filled catheter is inserted into the bladder. A 3D image of the prostate is created on the computer with the obtained flat ultrasonic sections.
- Preparing needles. Needles are supplied by manufacturers, preloaded, in sterile packaging, or, most often, as a thread containing radioactive sources. The doctor cuts these threads and loads the implant needles using a special instrument ‒ a needle loading station.
- Implanting the sources into the prostate through the perineal area. Needles are inserted through the grid holes with a stepping device. To prevent the gland from shifting, two fixing needles are placed. The needles are inserted by hand, starting from the bottom or top row of the coordinate grid. The depth of needle insertion is controlled by using ultrasound or fluoroscopy.
When high-dose rate brachytherapy is used, the needles are inserted with nylon catheters that are not removed.
Each needle has an echogenic tip which is displayed on the computer screen. When inserted, it must coincide with the planned point.
The radioactive seeds are first implanted into the anterior lobe of the prostate to avoid their further darkening in the posterior lobe.
The brachytherapy procedure for prostate cancer takes a little time ‒ 15-40 minutes. After the operation the patient radiation exposure is monitored. The equivalent dose rate should not be more than 10 µSv/ h at a distance of one meter from the patient. Depending on the patient’s condition, his discharge from the hospital is possible soon after the operation.
As there is some likelihood that radiation sources can go out of the body, it is recommended to use a bedpan or a urinal for urinating during the first 5 days after the operation or urinate through gauze, and to use condoms during sexual intercourse for 6 months.
Benefits of Brachytherapy
Does it affect urination?
After such therapy in clinics with little experience in brachytherapy or with obsolete equipment the patient can face a variety of complications like an urination disorder (urinary frequency, urinary retention or pain). Brachytherapy in German clinics ensures good results due to the vast experience of the specialists and low complication rates (one per cent out of 100%).
Influence on the sexual function
A healthy reproductive system is crucial to each man; therefore, even while treating such a serious disease as prostate cancer, many patients are worried about the impact of the therapy on their sexual function. Brachytherapy makes it possible to treat the disease with a minimal risk of sexual dysfunction. The erectile function usually remains unaffected after the therapy, but in case of unsatisfactory erection it can be improved by medications and other adjuncts during the therapy.
Brachytherapy in Germany and its impact on gastrointestinal function
Compared to other methods brachytherapy is the safest treatment of prostate cancer in Germany. Unlike beam-therapy, brachytherapy can seldom lead to intestinal upset. Due to its safety and effectiveness brachytherapy is a popular and a highly valued treatment in Germany.
Brachytherapy: Chances for Success
Brachytherapy in Germany is the safest and the most effective method to treat prostate diseases that ensures high survival rates, nearly 90%, among the patients. This treatment is similar to prostatectomy, though the complication rate is much lower after brachytherapy than after prostatectomy, for instance, urinary incontinence rates are lower than 4% and erectile dysfunction rates are lower than 15%. In few days or even in one day after brachytherapy patients can return to a normal way of life.
Side Effects of Prostate Brachytherapy
Brachytherapy is prescribed only when the pathogenic mass is localized, since radiation extends only 1-2 mm outside the microcapsule. The acceptable threshold for performing this treatment for prostate cancer is stages T1 and T3. Irradiation is not prescribed for men who have a pronounced urinary disorder, an increase in PSA more than 15-20 ng/ ml, and affected seminal vesicles.
In rare cases patients may experience the following complications:
- Urinary problems accompanied by sever colic, which indicates the development of inflammation provoked by the operation.
- Constriction of the urethra. The complication can cause acute urinary retention.
- Urinary incontinence. The percentage of this complication is very low.
- Erectile dysfunction. The probability of its development depends on the patient’s age. If a man had a high potency before the surgery, he will not have this complication.
- Inflammation of the rectum, one of the most serious complications. Small erosions appear on the inner surface of the organ, which, as a rule, leads to bleeding. This complication may be accompanied by itching and pain in the affected area.
- Migration of radioactive seeds. They may get into the bladder. The risk of such complications is very low.
If the surgery is performed by a qualified specialist, and the patient follows all the recommendations after it, there is no need to worry about the onset of complications.
Post-surgery Recommendations After Brachytherapy
On the day of checkout from the clinic, the power of radiation around the patient shouldn’t exceed 10 mcSv/h at the distance of 1 meter, which corresponds with German norms of radiation safety for the safety of people around. The medical physicist of the clinic where brachytherapy was performed should measure the value of radiation around the patient prior to letting him go from the clinic and write down these numbers in the checkout list. Down the road, this checkout list can be used for reference and explanation, if the patient will be stopped at detectors of metal and radiation in airports, industrial sites, etc. High radiation can be registered within 6 months after brachytherapy because the half-life is about 60 days. When a patient will be controlled with metal detectors, there’s a possibility that it will react, because a micro-source is a small metal capsule. Therefore, a patient should always have a copy of the checkout list and keep it nearby. For crossing the state border, a patient may need to have checkout list translated into English. It can be requested in the clinic, or a notarized translation can be made. Prior to leaving the clinic, a patient should get recommendations that will help him to avoid possible complications and will ensure the safety of family members:
- 2 weeks after the operation do not lift anything heavier than 5 kg. The load should be lifted carefully, without abrupt movements;
- don’t sit more than 1,5-2 hours in a row;
- obtain from physical exercises and load;
- keep tabs on intestine and bladder discharge, don’t let them overfill;
- avoid spicy, fried and hot food. Obtain from drinking beer;
- small children should not be seated on patient’s lap 2 months after the operation;
- pregnant women shouldn’t spend much time near the patient for too long 2 months after the operation.
There’s a possibility that the micro-source can get outside together with urine. It is especially applicable to patients who went through trans-urethral resection before brachytherapy. If a patient has managed to notice the moment of source discharge, it shouldn’t be touched with hands. The capsule should be taken with some device and placed in some container and stored away from children. When visiting a hospital, a patient should give the capsule to the doctors for recycling.
If a patient needs to go through treatment of another disease, before doing it (especially physical procedures), a person should consult with the oncologist who performed brachytherapy. During 2 years after the operation, it’s crucial to follow this recommendation.
In 1-1.5 months after implantation of micro-sources, a patient can get back to normal life, if the swelling of the prostate gland subsided, and it turned back to its previous size (it can be defined when a patient visits the doctor in 1-1.5 months after implantation). The weight of lifted load can be increased, a patient can visit swimming pool and start performing simple physical exercises. As the result, in 3-4 months, a person can totally get back to active social and physical life. In 3 months after the procedure, PSA (prostate-specific antigen) test should be made. In 6 months after the implantation, the process of isotope decay will be finished, and the patient will be safe for people around. During the first year, PSA test and ultrasound tests should be made every 3 months. During the following years, it should be done every 6 months.





































 Loading ...
Loading ...


